In a study published in the January 19, 2016 issue of the Journal of the American Chemical Society (JACS), scientists at Tsinghua University in China confirmed that something very unusual is happening inside extremely heavy atoms, causing them to deviate from their expect chemical behavior predicted by their place on the Periodic Table of Elements. Due to the velocity of electrons in these heavy elements getting so close to the speed of light, the effects of special relativity begin to kick-in, altering the chemical features observed.
The study shows that the behavior of the element Seaborgium (Sg) does not follow the same pattern as the other members of its group, which also contain Chromium (Cr), Molybdenum (Mo), and Tungsten (W). Where these other group members can form diatomic molecules such as Cr2, Mo2, or W2, using 6 chemical bonds, diatomic Sg2 forms using only 4 chemical bonds, going unexpectedly from a bond order of 6 to a bond order of only 4. This is not predicted by the periodic nature of the table, which itself arises from quantum mechanical considerations of electrons in energy shells around the nucleus. So what’s happening here? How does relativity throw off the periodic pattern seen in our beloved table of elements?
The Periodic Table of elements was initially conceived by Dmitri Mendeleev in the mid-19th century, well before many of the elements we know today had been discovered, and certainly before there was even an inkling of quantum mechanics and relativity lurking beyond the boundaries of classical physics. Mendeleev recognized that certain elements fell into groups with similar chemical features, and this established a periodic pattern to the elements as they went from light weight elements like hydrogen and helium, to progressively heavier ones. In fact, Mendeleev could predict the very specific properties and features of, as yet, undiscovered elements due to blank spaces in his unfinished table. Many of these predictions turned out to be correct when the elements filling the blank spots were finally discovered. See figure 1.
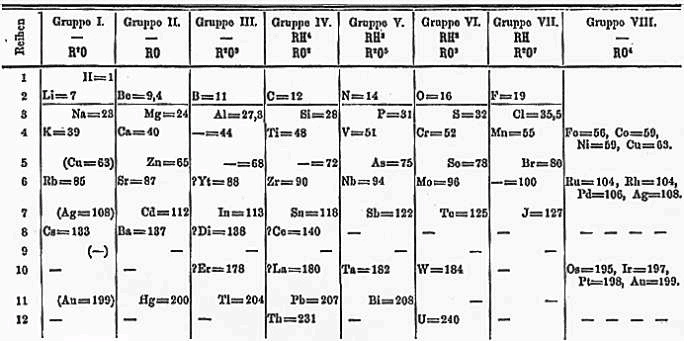
Figure 1. Mendeleev’s 1871 version of the periodic table. Blank spaced were provided where predicted new elements would be found.
Once quantum theory was developed in the early 20th century, the explanation for the periodic behavior of the table became apparent. The electrons in the atom are arranged in orbital shells around the nucleus. There are several different orbital types, again based on predictions from quantum mechanics, and each type of orbital can hold only a specified number of electrons before the next orbital has to be used. As you go from top to bottom in the Periodic Table, you use orbitals of progressively higher energy levels. Periodic behavior arrises because, although the energy levels keep getting higher, the number of electrons in each orbital type are the same for each group, going from top to bottom. See figure 2.
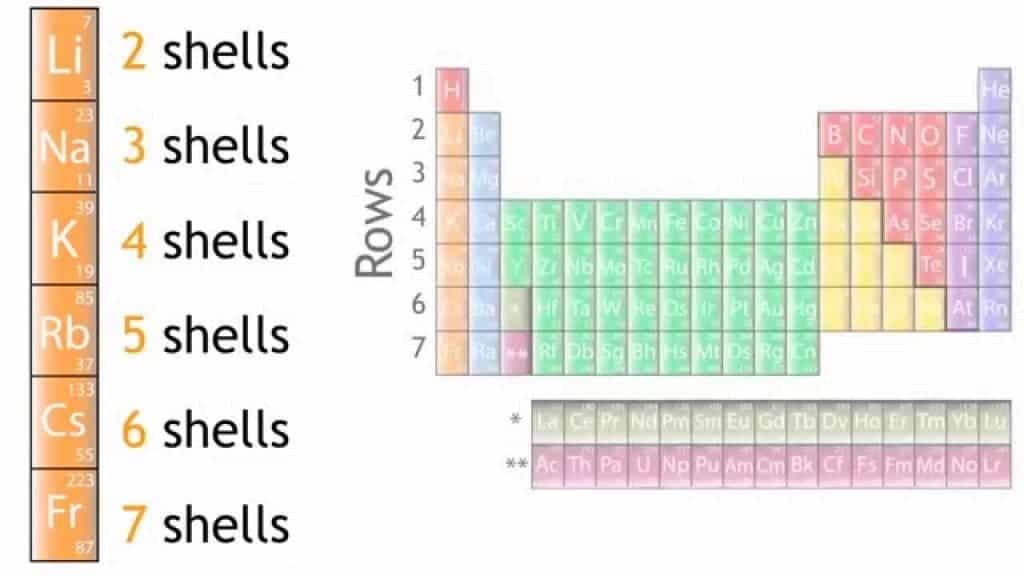
Figure 2. Group 1 as an example of a group in the Periodic Table. As the group goes from top to bottom the energy levels get higher and the elements get heavier.
The other great area of physics developed in the early 20th century was relativity, which didn’t seem to have much importance on the scale of the very small. Albert Einstein published his ground breaking paper on Special Relativity (SR) in 1905, which described the effects on an object moving close to the speed of light. In 1915 he developed the General Theory of Relativity (GTR), describing the effects due to a massive gravitational field. It is SR that becomes an important consideration in the very heavy elements due their electrons reaching velocities at a significant percentage of the speed of light.
Einstein showed that as the velocity of an object approaches the speed of light its mass increases. This effect is too small to be noticeable at everyday speeds, but becomes pronounced near light speed. It can also be shown that the velocity of an electron in orbit around an atom, is directly proportional to the atomic number of the atom. In other words, the heavier the atom, the faster its outer electrons are moving. For the element hydrogen, with atomic number 1, the electron is calculated to be moving at 1/137 the speed of light, or 0.73% of light speed. For the element gold (Au) with atomic number 79, the electrons are moving at 79/137 the speed of light, or 58% of light speed, and for Seaborgium (Sg) with atomic number 106, the electron is going at an impressive 77% of light speed. At these speeds the crazy effects of special relativity kick-in making the electron mass significantly heavier than it is at rest. For gold this makes the electron 1.22 times more massive than at rest, and for Seaborgium the electron’s mass comes out to be 1.57 times the electron rest mass. This, in turn, has an effect on the radius of the electron’s orbit, squeezing it down closer to the nucleus.
Some relativistic effects have already been known for certain heavy elements. The color of gold, for instance, arises due to the effects of relativity acting on it’s outer electrons, altering the energy spacing between two of it’s orbitals where visible light is being absorbed, and giving gold it’s characteristic color. If not for these relativistic effects, gold would be predicted to appear whitish.
For the elements in Group 6 of the Periodic Table (Cr, Mo, and W) (see Figure 3.) that were studied in the JACS article, they each have five d-orbitals and one s-orbital capable of forming bonds with another atom. Sg breaks the periodic pattern because it’s highest energy s-orbital is so stabilized by the effects of it’s relativistically moving electron, it doesn’t contribute to bonding. Due to the intricacies inherent in molecular orbital theory, this drops the number of bonding orbitals from 6 in Cr, Mo, and W, to only 4 in Sg (even though Sg is a group 6 member). It also means that the bond between Sg and Sg in the Sg2 molecule is 0.3 angstroms longer than expected, even though the Sg radius is only 0.06 angstroms bigger than W. If relativity didn’t have an effect, then the Sg2 molecule would be joined together by 6 orbital bonds, like any respectable Group 6 element should be! The same effect was also found in the Group 7 elements, with Hassium (Hs) showing the drop in bond order due to relativistic effects, just as Sg.
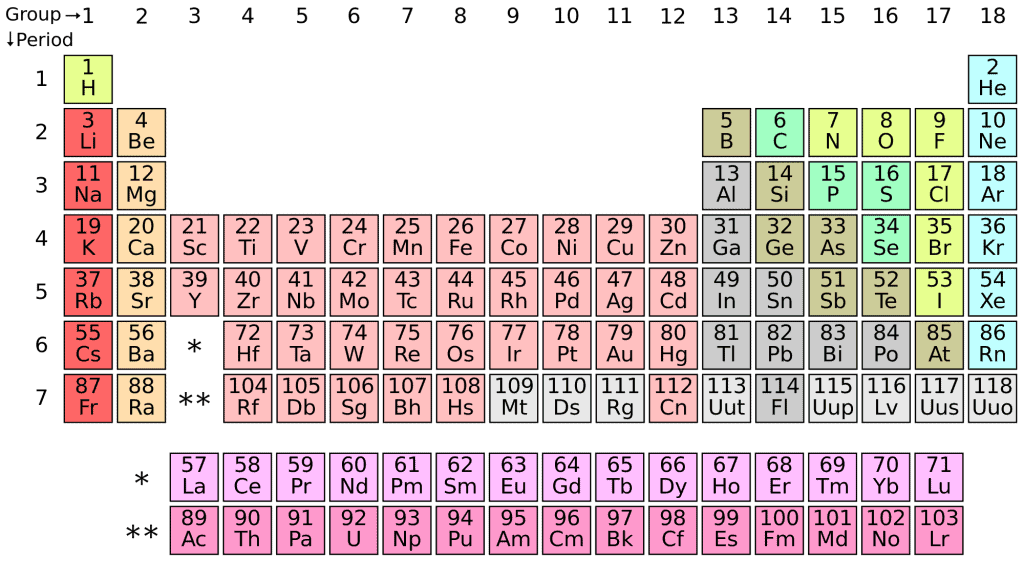
Figure 3. A modern version of the Periodic Table of Elements. Notice the Group 6 elements Cr, Mo, W, and Sg.
The periodic table of elements is an impressive scientific achievement, who’s periodicity reveals an underlying order in nature. While this periodicity works remarkably well, the few exceptions to the rule also uncover important principles at work. Einstein’s theory of relativity breaks the periodic table in some interesting and unexpected ways. It’s the very heavy elements on the chart that don’t show good “table” manners, thanks to Einstein.
Journal Reference and other reading:
1. Relativistic Effects Break Periodicity in Group 6 Diatomic Molecules Yi-Lei Wang, Han-Shi Hu*, Wan-Lu Li, Fan Wei, and Jun Li*
Department of Chemistry & Key Laboratory of Organic Optoelectronics and Molecular Engineering of Ministry of Education, Tsinghua University, Beijing 100084, China J. Am. Chem. Soc., 2016, 138 (4), pp 1126–1129 DOI: 10.1021/jacs.5b11793 Publication Date (Web): January 19, 2016
2. Relativistic effects in structural chemistry Pekka Pyykko Chem. Rev., 1988, 88 (3), pp 563–594 DOI: 10.1021/cr00085a006 Publication Date: May 1988
3. Why is mercury liquid? Or, why do relativistic effects not get into chemistry textbooks? Lars J. Norrby J. Chem. Educ., 1991, 68 (2), p 110
DOI: 10.1021/ed068p110 Publication Date: February 1991









